Published: April 29, 2026 | Published By: Real Creative Agency
Charlie’s Holdings OTCQB:CHUC is an industry leader in the premium vapor products space with more than 650 active Premarket Tobacco Product Applications (PMTAs) with the FDA.
To provide adult smokers with better alternatives to combustible cigarettes, Charlie’s has developed a family of award-winning e-liquids as well as an array of compact, easy-to-use disposable vaping devices.
The Company’s products are sold around the world to select distributors, specialty retailers, and third-party online resellers. Many store owners report that Charlie’s vapor products are among their fastest growing offerings.
CHUC stock is up more than 600% over the last 13 months.
Based on the Company’s competitive advantages, age-gating license, and plans for a 2026 up-list to a national securities exchange, we think that CHUC is a VERY RARE find in the penny stock world.
Here is a profitable penny stock, with rapidly growing revenue, consistent insider buying, and focused on uplisting to a major exchange.
In March, CHUC hosted investor meetings at the Roth Conference at The Ritz-Carlton Laguna Niguel in Dana Point, California.
As one of the largest investment conferences for small-cap companies, the invitation-only event connected executive teams from roughly 500 private and public growth companies with institutional investors.
After the conference, I sat down with Henry Sicignano, CHUC President, to learn more about the Company’s strategic plans for 2026.
Scott Shaffer: The combination of monetizing a tiny number of the Company’s PMTAs and the launch of nicotine category killer SBX delivered a fantastic 2025.
Now CHUC has four compelling categories of competitive advantage:
- a massive PMTA portfolio;
- the upcoming FIRST AI age-gated flavored vape in the United States;
- nicotine category killer SBX;
- the Company’s new US manufacturing facility.
Can you give a quick summary of each of these “strategic moats”?
Henry Sicignano:In a market rife with illegal, non-compliant, and irresponsibly marketed products, Charlie’s has worked tirelessly to forge competitive advantages built around regulatory compliance and responsible business practices.
And now, after six years of investment, we believe that Charlie’s is emerging as a true industry leader in the US vapor products industry.
Here are the primary areas that distinguish us:
1. PMTA Product Portfolio. In 2025, in three separate transactions, Charlie’s sold sixteen of the Company’s 694 PACHA synthetic nicotine PMTA products to one of the world’s largest tobacco companies for $7.5 million cash plus a contingent one-time payment of up to $4.2 million.
In the last of the three transactions, the buyer purchased a single Charlie’s PMTA product for $1 million.
Based on these sales, taking into account the fact that Charlie’s continues to own 678 PMTA products, and considering the interest other companies have expressed in Charlie’s portfolio, the Company believes Charlie’s remaining PMTA products, as a stand-alone asset, have a monetary value that far exceeds Charlie’s current market cap.
To maximize the value of this portfolio, the Company intends to:
- continue to amend and strengthen Charlie’s PMTAs with new scientific data:
- add age-gating functionality to certain of our PMTAs
- explore new strategic partnerships with industry competitors, big and small, that value regulatory compliance in the vapor products marketplace.
2. Age-Gated Flavored Products. Currently, there is a need for age-gated product technologies that can satisfy concerns the FDA has related to underage youth access.
We believe age-gating is both a responsible business practice as well as a potentially massive competitive advantage for Charlie’s. The Company also believes that effective age-verification mechanisms will be a critical component in supporting the Company’s PMTAs for both flavored and “plain” tobacco nicotine vapor products.
Utilizing the Company’s December 2025 licensing agreement with IKE Tech LLC for the first-ever AI-powered blockchain-based age-gating system for vapor products in the United States, in mid-2026 Charlie’s plans to test-market the patented IKE age-gating system with one or more special lines of the Company’s popular disposable brands.
Under the IKE license, Charlie’s could become the first Company to demonstrate to the FDA that flavored ENDS products are “appropriate for the protection of public health.”
Such a regulatory achievement could prove transformative for Charlie’s and for the entire industry.
3. Non-Nicotine SBX Products. Our market research indicates that adult consumers overwhelmingly prefer “flavored” vapor products over plain tobacco products and are highly receptive to nicotine substitute products that offer the same vaping experience as that provided by conventional nicotine vapor products.
Because the FDA has not yet issued marketing orders for any flavored nicotine vapor products, we believe that non-nicotine SBX Disposables are a critical pillar of Charlie’s product portfolio while we continue to strengthen and amend our PMTAs for flavored nicotine products.
SBX nicotine analogue-based vape liquids are not made from or derived from tobacco, nor do they contain nicotine from any source.
Accordingly, the Company’s proprietary nicotine substitute alkaloid (patented in the United States and in China by the Company’s chemical supplier and trademarked by Charlie’ as Metatine™) does not meet the definition of “nicotine.”
Accordingly, SBX products are not subject to FDA PMTA requirements and are LEGAL across most of the United States.
Some states have begun passing legislation to ban certain flavored nicotine products. In these select states, where flavored nicotine products are now beginning to face significant regulatory restrictions, we are focusing our non-nicotine SBX sales initiatives.
We believe that the list of states that give SBX a “regulatory advantage” will grow.
Given that Charlie’s SBX Disposables are overwhelmingly preferred over Juul tobacco-flavored vapes (as highlighted by our Company-sponsored focus group survey), if SBX is able to achieve just a fraction of Juul’s retail distribution, Charlie’s sales will grow impressively in “advantaged states” across the country.
4. U.S. Manufacturing. In Q4 2025, the Company opened an Electronic Nicotine Delivery System (“ENDS”) filling operation in the United States.
The establishment of this domestic operation reduces production costs, mitigates supply chain disruptions associated with importing finished goods, and enables Charlie’s to meet stringent domestic manufacturing requirements of Texas and other large states.
The Company’s US Manufacturing facility also enhances the appeal of Charlie’s premium products, broadly, to adult consumers who prefer “Made in America” brands and represents, we believe, an important competitive advantage going forward.
Scott Shaffer: Which “strategic moat” excites you the most, and which ones do you think Big Tobacco is keeping an eye on?
Henry Sicignano: Charlie’s PMTA Product Portfolio – in combination with our age-gating initiative – represents Charlie’s greatest opportunity… and certainly an initiative of high interest to Big Tobacco.
Adult consumers overwhelmingly prefer (by more than 4:1) “flavored” vapor products over plain tobacco products and yet, to date, the FDA has not “approved” a single flavored nicotine vapor product. In this environment, all the big tobacco companies are keenly interested in whatever product that might show promise in winning an FDA marketing order.
Charlie’s believes that an effective age-verification mechanism – paired with a flavored ENDS product under a strong brand – will be a critical component in supporting the Company’s PMTAs for flavored nicotine vapor products.
Under the IKE license, Charlie’s could become the first Company to demonstrate to the FDA that flavored ENDS products are “appropriate for the protection of public health.”
Such a regulatory achievement would be a game-changer for Charlie’s and for the entire vapor products industry.
Big tobacco would certainly take note of such an achievement.
Scott Shaffer: Charlie’s PMTA sales/licensing activity seems to have quieted down; any reason why? What should we expect going forward?
Henry Sicignano: After Charlie’s 2025 PMTA asset sales, the Company and our counsel fielded numerous inquiries regarding our remaining 678 PMTA Products.
But licensing transactions are complicated and the FDA’s regulatory position has been highly inconsistent.
Just this month the Nicotine Insider reported that internal FDA memos show that Agency scientists backed the authorization of a California company’s flavored nicotine pods… but senior FDA leadership blocked the authorization. In this confusing regulatory environment, Big Tobacco companies are understandably cautious.
That said, Charlie’s remains in discussions around licensing possibilities with three companies.
We believe, strongly, that when Charlie’s introduces age-gated disposables to the market we will have our pick of Big Tobacco licensee candidates.
Scott Shaffer: Is the goal still to amend some or all the PMTAs to include age-gating technology?
Henry Sicignano: Though our long-term goal would be to offer at least dozens – and perhaps hundreds – of flavored vapor products to adult consumers, PMTAs are expensive. And time consuming. That is why, at present, we intend to start with 10-15 flavored products.
Scott Shaffer: SBX is now in AR, KY, MS, LA, NY, NC, VA and the first and only 25K puff legal to sell in California. What other states are you targeting?
Henry Sicignano: As the 2026 state budget and legislative sessions wind down, new regulations impacting the availability of nicotine products will take effect. State restrictions on electronic nicotine delivery systems, or “ENDS” products, create excellent opportunities for SBX.
As you mentioned, SBX recently launched in California to strong demand from retailers and distributors. Pennsylvania and South Carolina are two other states with nicotine restrictions taking effect before the end of 2026. We have also seen the success and demand for SBX bleed into states surrounding nicotine restricted markets.
And, just to be clear, in some states that have posed restrictions on ENDS products, SBX isn’t the only Charlie’s product that has achieved regulatory success… PACHA 10K, 15K, and 20K-puff Virginia Tobacco disposables are also legal to sell in California!
Scott Shaffer: When the Government shutdown last year and the FDA stopped cracking down on illegal vapes, once again the Chinese flooded the US market with illegal vapes. How does this problem get solved?
Henry Sicignano: That is an excellent question. And I am not sure I have an answer. We are strong proponents of regulatory enforcement and the establishment of a truly level playing field for all players in the US vape industry.
For years consumers have been purchasing BILLIONS OF DOLLARS, ANNUALLY, of illicit Chinese vapes in the United States. Last year’s FDA/state crackdowns on illicit products significantly benefited Charlie’s – and our SBX product line, in particular.
Unfortunately, since the beginning of January we have seen very little enforcement of existing vape product laws. What’s more, the largest Chinese manufacturers of illicit vape products “dumped” more than six months’ worth of inventory in the United States. Simultaneously, to crush American competition, the Chinese drastically reduced prices.
By “loading up” the US trade with billions of dollars of illicit product – with severe, predatory pricing – Chinese competitors have certainly hurt US companies.
It is our hope that:
- state regulatory enforcement will step up in the second half of 2026,
- the FDA will provide a legitimate PMTA pathway for the authorization of flavored ENDS products
- that Charlie’s age-gating initiative will successfully demonstrate to the FDA that our flavored ENDS products are “appropriate for the protection of public health.”
Scott Shaffer: Tell us more about CHUC’s new 75K puff vapes.
Henry Sicignano: Charlie’s is very excited about Pachamama 75K and SBX 75K disposables.
For more than a year, our R&D team worked closely with our manufacturing partners to develop proprietary 75,000-puff devices that utilize best-in-class technology, Charlie’s award-winning flavors, and incredibly elegant casings.
The result?
Exceptional next generation products that we know will delight Charlie’s customer base.
Over the last 30 days we have begun to release Charlie’s 75,000-puff products to a small number of our retail and distributor partners.
Early customer feedback has been excellent… and indicates user experience is as good – or better than – the experience provided by industry leading (and illicit) Foger and Geek Bar brands.
As we expand our distribution, Pachamama 75K and SBX 75K are well-positioned to find strong success throughout the remainder of 2026.
Scott Shaffer: In March the FDA issued new draft guidance for flavored vape products. Can you give us your thoughts? Does an FDA approved age-gated vape negate the need for a PMTA for the solution inside?
Henry Sicignano: As noted in the Draft Guidance, prior to marketing a new vape product, manufacturers must obtain FDA authorization and demonstrate their product marketing aligns with FDA consideration as to whether the marketing and sale of the products is “appropriate for the protection of the public health.”
The FDA’s PMTA review process remains unchanged, requiring applicants to clear a very high evidentiary burden and must show the benefits (i.e., aiding smoking cessation) outweigh potential risks to the population as a whole, particularly the risk of youth initiation.
To date, out of 27 MILLION PMTAs, nearly all ENDS products have failed to earn a “marketing granted order” from the FDA.
In fact, FDA has only approved 39 ENDS products (all in tobacco and menthol flavors) from large tobacco manufacturers.
As I explained above, under the IKE license that will facilitate America’s first age-gated flavored ENDS, we believe that Charlie’s could become the first Company to demonstrate to the FDA that flavored ENDS products are “appropriate for the protection of public health.” Such a regulatory achievement would be a game-changer for Charlie’s and for the entire vapor products industry.
Scott Shaffer: It sounds like your partnership with IKE Tech and plans for America’s first AI-powered age-gating vape are extremely important to Charlie’s. When do you expect the first age-gated flavor vape to launch? Will it be with a PMTA product or SBX?
Henry Sicignano: Charlie’s pioneering age-gating initiative really is extraordinary. Representing an industry first in deploying proprietary technologies that protect public health, enable regulatory compliance, and have the strong potential to drive significant industry growth, our product could be game-changing. So we are VERY eager to launch!
That said, we want to make sure that everything about the product – and everything surrounding our launch – are very carefully considered.
Accordingly, though we had initially planned to launch at the start of summer with our SBX nicotine analogue products, we are now planning a Q3 2026 test-market of the patented age-gating system in a special line of the Company’s popular PACHA brand disposables.
We will likely launch the product in 200-300 stores this August and plan to simultaneously begin amending our corresponding PMTAs with the FDA.
Scott Shaffer: Why would a retailer choose to stock Charlie’s first-of-a-kind, age-restricted product if it will need to compete against established illegal vapes? Do you have a specific convenience store chain or two in mind that are compliant?
Henry Sicignano: There is a large un-met need for technologies that can satisfy or accommodate concerns the FDA has related to youth access… which means there is a multi-BILLION market opportunity for flavored vapes that are inoperable for underage individuals.
That said, unless the FDA – or individual state regulators – begin to enforce existing laws and regulations, “mom & pop” retailers who currently carry illicit products likely will not have interest in Charlie’s age-gated PACHA products.
But, keep in mind, there are more than 100,000 national and regional chain convenience stores across the United States that are committed to regulatory compliance and therefore do not carry illicit products.
Currently offering only FDA-authorized tobacco-flavored products, responsible U.S. retailers are desperate to be able to offer FLAVORED vapor products that 85% of adult ENDS consumers prefer.
Further, when regulatory enforcement does (eventually) impact the independent “mom & pops,” we believe that the vast majority of all US retailers will begin to carry only age-restricted vapor products.
Scott Shaffer: In the marketplace, if given the choice, will consumers purchase age-gated flavored products over non-restricted tobacco-flavored vapes?
Henry Sicignano: Both the e-cigarette industry and public health officials have come to recognize that 80-90% of adult consumers strongly prefer flavored vapor products over tobacco, menthol, or “clear” flavors and understand that most adult cigarette smokers will continue using traditional combustible products if flavored vapor products are not available.
California, Massachusetts, parts of New York State, and many other regions across the country have also imposed significant restrictions or outright bans on flavored ENDS products. These bans have led to an explosion of illicit foreign products, confusion among retailers, and, most unfortunately, to the elimination of legal flavored products as an option for many adult cigarette smokers who would prefer to switch to far less harmful (flavored) vapes.
In this environment, we believe that if an adult consumer is given the choice between a conventional tobacco-flavored product vs. an age-restricted FLAVORED product, the overwhelming majority will choose the age-restricted product.
Henry Sicignano: We believe that the market opportunity for age-restricted flavored ENDS products exceeds $6 billion annually.
Scott Shaffer: Once you launch the age-gated flavored product, what is your intention? Would you amend more PMTAs to include age gating?
Henry Sicignano: Once we initiate our 200-300 store market test of our age-gated PACHA products, we intend to collect market data around sales velocity, consumer demographics, ease of use, and consumer intent to re-purchase.
We intend to share this information with the FDA and to initiate what we hope will be collaborative dialogue with the Agency.
We plan to engage in discussions with certain of our largest competitors.
If one or more of these efforts prove fruitful, we think it highly likely that strategic partnership(s) will emerge.
Then, with deep pocketed partners, we would certainly endeavor to write age-gating functionality amendments for dozens (or even hundreds) more of our 678 PMTAs.
Scott Shaffer: Charlie’s currently owns 678 PMTAs… and completed its last sale of a single PMTA for $1 million…
If you are able to launch an age-gated flavored vape under one of Charlie’s timely-filed PMTAs, should that make Charlie’s PMTA portfolio worth MORE than today’s imputed value of $678M?
At what point do you think Big Tobacco starts to get interested in CHUC and/or the Company’s PMTAs?
Henry Sicignano: The best way to forecast the value of Charlie’s PMTA products might be to survey asset sales of comparable products. Setting aside the 2018 anomaly that was Altria’s purchase of 35% of Juul for $13 billion, I think it might be useful to consider another, relatively more “reasonable” comparable…
In 2023 Altria purchased NJOY for $2.75 billion. This acquisition included (only) 5 TOBACCO-flavored PMTA skus.
Meanwhile, Charlie’s has quietly amassed a portfolio of HUNDREDS of FLAVORED PMTA-submitted skus (in addition to more than 10 tobacco-flavored skus). Admittedly none of Charlie’s PMTAs has yet received an FDA marketing order.
Still, even in their current review status with the FDA, in 2025 one big tobacco company was willing to purchase a single ONE of Charlie’s PMTA products for $1 million.
After selling 16 of our PMTA skus to this particular partner, Charlie’s has 678 remaining.
Going forward, our intention is to sign evergreen LICENSING deals with other large industry partners.
What might a small set of these products, say 10-15 skus, be worth in a sale once they are modified to include age-gating technology?
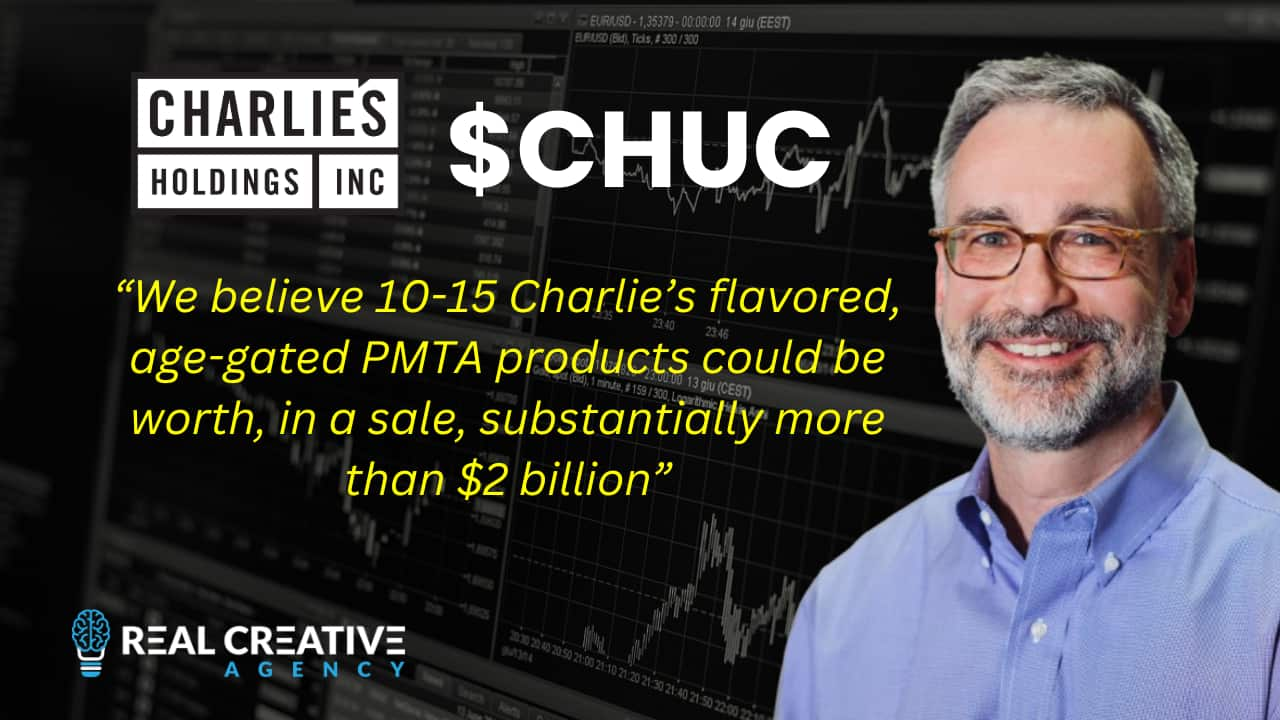
Given that adult consumers overwhelmingly prefer flavored products (by a margin of greater than 4:1), and given that Altira paid $2.75 billion for only 5 (far) less desirable tobacco flavored skus, we believe 10-15 Charlie’s flavored, age-gated PMTA products could be worth, in a sale, substantially more than $2 billion… or more than 25X today’s share price.
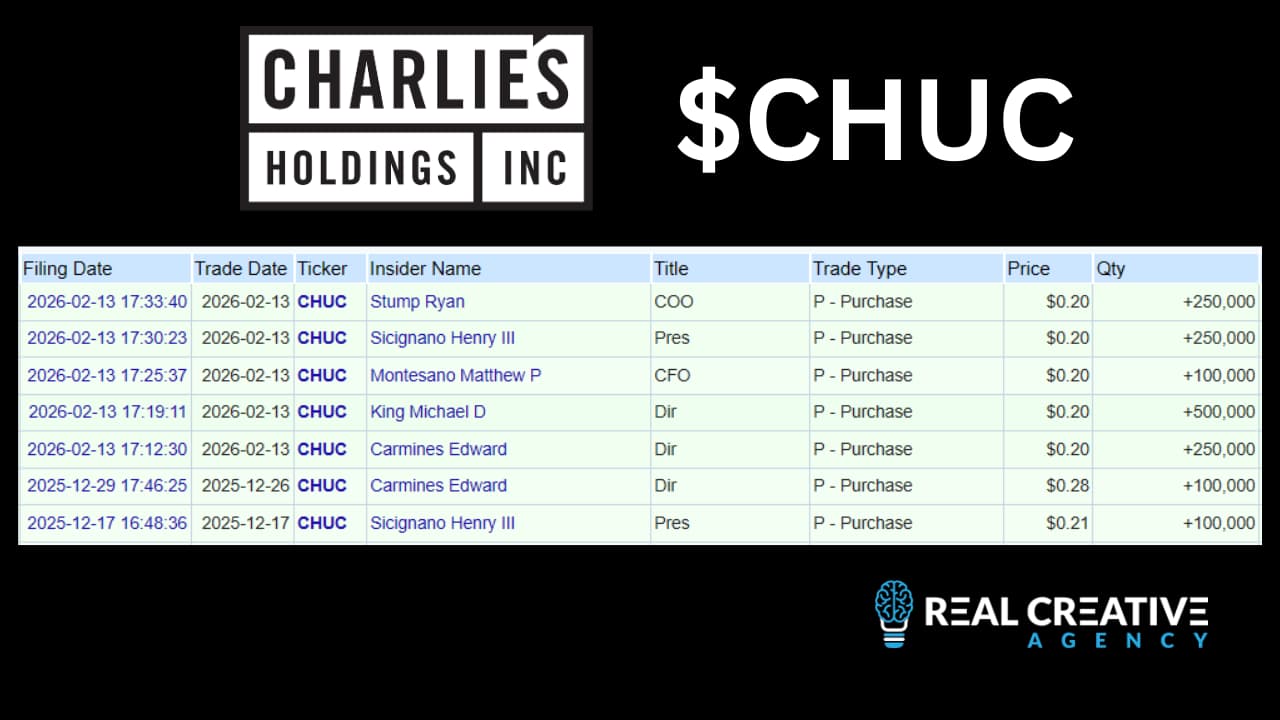
This is one of the reasons company insiders and founders have been consistent buyers of CHUC shares on the open market… and why a few of us invested $270,000 in the last funding round.
Scott Shaffer: Reynolds seems to be doing well with VUSE brand vapor products (though they still have no flavored vapes in the US market)… and Altria purchased NJOY in 2023. Which of these Big Tobacco behemoths might be the most logical strategic partner for CHUC?
Henry Sicignano: Unfortunately for Altria, the U.S. International Trade Commission ruled that Altria’s NJOY devices and pods infringed on patents held by Juul Labs. Accordingly, the importation and sale of NJOY products were banned on March 31, 2025.
This ban remains in effect today, leaving Altria without a vapor product in the United States.
Under these circumstances, compared with big tobacco competitors, Altria is losing the smokeless tobacco race. Badly.
While Altria is sitting on the vapor products sideline, Reynolds’ VUSE e-cigarettes are generating more than $1 billion in the lucrative US market.
For these reasons, it seems obvious that Altria should have a (very) high degree of interest in Charlie’s and our PMTA portfolio.
As I mentioned, we plan to launch a number of age-gated, FLAVORED PMTA products in 200-300 stores this August… and will simultaneously begin amending our corresponding PMTAs with the FDA.
As soon as we can compile market data from this test, we believe it likely that Altria – and other Big Tobacco companies – will have commercial interest in our products.
Scott Shaffer: Your auditors have removed their “going concern” opinion from the Company’s financial statements; does this achievement bode well for Charlie’s plans to up-list to a national securities exchange?
Henry Sicignano: 2025 was a fantastic year for Charlie’s. If one were to include, as top-line revenue, our $7.5 million in PMTA product sales and our $3+ million in discontinued Don Polly sales, Charlie’s revenue actually eclipsed $30 million… and was the highest grossing year in company history!
As a result, our 2025 audit report no longer includes a ‘going concern’ explanatory paragraph, underscoring the substantial progress we have made in strengthening our financial position and liquidity.
With our strengthened financial position – combined with the Company’s decision to discontinue Don Polly LLC operations and to no longer sell Don Polly’s CBD/hemp branded products – we are now well-positioned to up-list to a national securities exchange… and we believe a 2026 up-list will significantly benefit Charlie’s shareholders.
Scott Shaffer: Charlie’s insiders and management have been big buyers of CHUC stock on the open market and in the latest funding… you guys have a ton of skin in the game.
Tell us why you LOOK FORWARD to a reverse split.
Henry Sicignano: Charlie’s Management, Directors, and affiliates currently own nearly 50% of the Company’s total outstanding shares.
With the closing of February’s private placement, CHUC Directors and members of management amassed an additional 1,350,000 shares – at a purchase price of $.20/share, with no warrants or any other inducements.
The common-only offering was exactly the kind of company-friendly equity raise that sets Charlie’s apart from other microcaps… and illustrates exactly how bullish CHUC insiders really are.
That said, Charlie’s insiders are keenly aware of the milestones the Company needs to achieve to up-list to a national securities exchange.
Since the beginning of 2026, the NYSE American has amended its minimum share price listing standards. As a result, the NYSE American and the Nasdaq now share a $4 share price as a minimum initial listing standard.
Given that Charlie’s currently has approximately 295 million outstanding shares (fully diluted), a $4 share price equates to a $1.2 billion market cap (compared with the Company’s current market cap of about $75 million).
While we DO believe that a Billion ++ market cap will ultimately be achievable, in order to increase liquidity , visibility, and the likelihood of analyst coverage in the very near term, we believe that achieving a $4++ share price through a reverse split — in advance of the Company achieving a $1.2 billion dollar market cap — is a strategic imperative that will be hugely beneficial to CHUC shareholders.
As the largest CHUC shareholders, Company insiders’ self-interests are squarely aligned with the interests of all our common shareholders.
Though we are cognizant of the reservations some investors have toward reverse splits, it is our belief that market cap – not share price – is the true measure of a company’s worth.
If the market believes that our company is worth $75MM, $150MM, or $300MM with 300 million shares outstanding… the market should believe that CHUC will have exactly the SAME market value with only 6, 10, or 15 million shares outstanding.
What’s more, because CHUC will be launching its reverse split “offensively,” to achieve minimum initial listing standards (as opposed to “defensively,” to remain above the Nasdaq’s minimum share price for remaining on the exchange), we believe that CHUC’s reverse split will be beneficial to common shareholders.
And, finally, because CHUC has no outstanding warrants or convertible debt, “fast money investors” will not be able to hedge short sales of CHUC common stock… so, unlike the catastrophes that befall companies like XXII, the circumstances surrounding CHUC’s up-list should be very positive.
With market-leading products designed for adult consumers, a clean balance sheet, and insiders with a TON of skin in the game, Charlie’s checks all the important boxes for an up-list to a national securities exchange.
We expect 2026 will be a very exciting year for our shareholders!
Other Articles About Charlie’s Holdings OTCQB:CHUC
- OTCQB:CHUC Best Nicotine Vape Stock
- Why CHUC Could Be A Must Own Penny Stock
- This Quiet FDA Decision Could Create A Billion Dollar Penny Stock
- Best Profitable Penny Stock with Insider Buying
- Big Tobacco Quietly Acquires CHUC PMTAs
- OTCQB:CHUC Investor Deck
- CHUC’s Latest Shareholder Letter
Safe Harbor Statement: This interview contains “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including but not limited to statements regarding the Company’s overall business, existing and anticipated markets and expectations regarding future sales and expenses. Words such as “expect,” “anticipate,” “should,” “believe,” “target,” “project,” “goals,” “estimate,” “potential,” “predict,” “may,” “will,” “could,” “intend,” variations of these terms or the negative of these terms, and similar expressions, are intended to identify these forward-looking statements. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. The Company’s actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to: the Company’s ongoing ability to quote its shares on the OTCQB; whether the Company will meet the requirements to up-list to a national securities exchange in the future; the Company’s ability to successfully increase sales and enter new markets; whether the Company’s PMTA’s for its nicotine-containing products will be authorized by the FDA, and the FDA’s decisions with respect to the Company’s future PMTA’s for nicotine products; the Company’s ability to manufacture and produce products for its customers; the Company’s ability to formulate new products; the acceptance of existing and future products; the complexity, expense and time associated with compliance with government rules and regulations affecting nicotine, synthetic nicotine, and products containing nicotine substitutes; litigation risks from the use of the Company’s products; risks of government regulations; the impact of competitive products; and the Company’s ability to maintain and enhance its brands, as well as other risk factors included in the Company’s most recent quarterly report on Form 10-Q, annual report on Form 10-K, and other SEC filings. These forward-looking statements are made as of the date of this interview and are based on current expectations, estimates, forecasts and projections as well as the beliefs and assumptions of management. Except as required by law, the Company undertakes no duty or obligation to update any forward-looking statements contained in this interview as a result of new information, future events or changes in its expectations.
Artificial Intelligence Stock Reading
- The Best Artificial Intelligence Penny Stock VERSES AI
- Artificial Intelligence Stock with BIGGEST Potential
- What is BETTER than ChatGPT Generative Artificial Intelligence
- Generative Artificial Intelligence Regulation Solution
- How To Govern and Regulate Artificial Intelligence
- The BEST C3 AI Stock Alternative
- Next Generation Intelligence Patent Application
- The ONLY Adaptive Artificial Intelligence Stock
- Artificial Intelligence Stocks
- VERSES VRSSF 2023 Year End Summary
- The Story of VERSES AI VRSSF
- How VERSES AI Solves the Generative AI Problem
Disclaimer
This communication is a paid advertisement for Charlies Holdings. to enhance public awareness of the Company, its products, its industry and as a potential investment opportunity. This communication is not intended as, and should not be construed to be, an offer to sell or a solicitation of an offer to buy any security.This communication is a paid advertisement for Charlies Holdings to enhance public awareness of the Company, its products, its industry and as a potential investment opportunity. Real Creative Agency, and their owners, managers, employees, and assigns were paid by the Company to create, produce and distribute this advertisement. This compensation should be viewed as a major conflict for this presentation to be unbiased.On August 7, 2025, Charlies Holdings agreed to pay Scott Shaffer (i) $5,0000 per month for 6 months (ii) issue 300,000 restricted shares of Charlies Holdings (CHUC).This communication is not intended as, and should not be construed to be, an offer to sell or a solicitation of an offer to buy any security. Neither this communication nor the Company purport to provide a complete analysis of the Company or its financial position. The Company is not, and does not purport to be, a broker-dealer or registered investment adviser. This communication is not, and should not be construed to be, personalized investment advice directed to or appropriate for any particular investor. Any investment should be made only after consulting a professional investment advisor and only after reviewing the financial statements and other pertinent corporate information about the Company. Further, readers are advised to read and carefully consider the Risk Factors identified and discussed in the government filings. Investing in securities is speculative and carries a high degree of risk.
Real Creative Agency
Digital Marketing Experts
Daily helping our most valuable asset "YOU" since 2005.